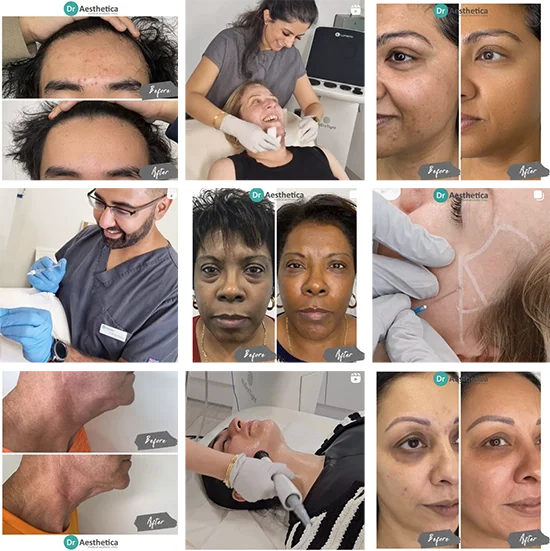
In the decades since its introduction, Botox has quite literally changed the face of humanity. In the UK, approximately 100,000 Botox injections are carried out each year. While technically a brand name, Botox may refer to any injectable form of botulinum toxin type A—a protein derived from the bacterium Clostridium botulinum. Other common brand names include Dysport/Azzalure, Xeomin/Bocouture, and Jeuveau (in the US and UK, respectively).
You Might Also Enjoy Reading: Azzalure Vs. BOTOX®: Which one Is Best For You
Botulinum toxin has a long history that stretches all the way to the 1820s when a German scientist discovered the first strains of the toxin in spoilt sausages. Then at the turn of the 20th century, another doctor uncovered seven strains of botulinum toxin, four of which were harmful to humans.
It took almost another century before researchers discovered that injecting small amounts of one of the strains (Botulinum toxin type A) into hyperactive muscles relaxed them. In 1989, the first FDA approval for Botox was issued to treat crossed eyes and eye twitching. Back then, it was called Oculinum until the drug maker Allergan acquired it and renamed it Botox.
However, the real breakthrough for the cosmetic use of Botox was made in 1987 by two married Canadian doctors, who accidentally discovered the wrinkle-fighting properties of the toxin after noticing that patients who were receiving injections for facial spasms were also losing their frown lines. The demand for wrinkle smoothing with Botox short through the roof in the 90s. Back then, all cosmetic uses of Botox were Off-Label, and the first FDA approval didn't come until 2002.

Approved Uses of Botox in the UK
When a drug is approved—by the Medicines and Healthcare products Regulatory Agency ( MHRA ) in the UK—it means the regulatory body has reviewed the results from all manufacturer testing done on the medication or treatment and has granted approval for its use for a specific purpose. Simply put, the drug has been deemed safe enough that the benefits are greater than the potential risks when considering the intended use.
BOTOX® Cosmetic
The FDA first approved Botox for cosmetic use in 2002 for the treatment of glabellar lines ("eleven" lines between the brows). The regulatory body also requested the product be marketed as BOTOX® Cosmetic to distinguish aesthetic from therapeutic uses.
Since that initial FDA approval, Botox has been approved for use in 95 countries and has become the topmost non-surgical cosmetic treatment available for wrinkles and fine lines.
Some of the approved Botox cosmetic treatments in the UK include
- Excessive Underarm Sweating-Botox has been proven to be very effective in treating Hyperhidrosis and putting an end to the often embarrassing appearance of sweat-soaked/stained clothes.
- Facial wrinkles such as frown lines, "crow's feet" and forehead wrinkles.
At Dr Aesthetica, we offer the approved Botox Cosmetic treatments for the conditions mentioned above.
BOTOX® Therapeutic
Botox Therapeutic has a different goal in mind. Instead of targeting crow's feet, frown lines, and other wrinkles on your face, these therapeutic injections can directly address medical conditions such as chronic migraines, severe spasms affecting the neck and shoulder areas and overactive bladder.
Some of the approved Botox Therapeutic treatments in the UK include:
- Chronic migraine- effective treatment for patients with at least 15 headache days a month, with at least eight of those featuring migraine symptoms.
- Teeth grinding, or bruxism can be treated with botox.
- Upper limb spasticity-a common motor dysfunction arising from stroke and traumatic brain injury.
- Overactive bladder with symptoms of urinary incontinence
- Crossed-eyes, which is one of the earliest medical approvals for Botox.

At Dr Aesthetica, we only offer Botox Therapeutic treatments for Chronic Migraines.
What Does Off-Label Mean?
Once a drug is approved by FDA or MHRA for one medical condition, doctors are not prohibited from prescribing it for any medical issue they think it could benefit, regardless of whether it's been proven to work for that condition.
This use is often based on a thorough evaluation of the patient's needs. Unapproved use of an approved drug is often called "off-label", and it's common practice in medicine.
Since doctors go through almost a decade of medical training, they're deeply versed with the anatomy of the face — where the nerves are, what areas to avoid and so on. Based on this knowledge, it's common for doctors to extrapolate information from one clinical trial and apply it to a new use scenario.
This has, in turn, made off-label use of Botox fairly common. In fact, it's the off-label use of Botox that has helped it explode into a cultural phenomenon and medical triumph–increasingly being drafted for problems that go far beyond the cosmetic.
All the same, it's always important to discuss your goals with an accredited aesthetics doctor before determining the best use of Botox. Depending on your goals and your doctor's evaluation, you may be a great candidate for the FDA-approved uses of Botox, or you may be a candidate for off-label use. Click here to talk to an experienced Botox injector.

Common Off-Label Uses of Botox
- While Botox is only approved for anti-wrinkle injections around the eyes and on the forehead, thousands of doctors across the world used it to fight wrinkles on other areas of the face. These areas include nose wrinkles and a pebbled chin.
- While Botox has been approved for excessive underarm sweating, doctors are also successfully using it for off label treatment of overly sweaty hands and feet.
- Botox has been shown to alleviate Bruxism (painful teeth grinding) significantly. This involves injecting Botox into the masseter muscle. The same procedure can be used for non-surgical jaw reduction for people with overly broad square jaws.

At Dr Aesthetica, we offer Botox Off-Label treatments for all the conditions mentioned above.
Potential Uses of Botox Still Under Clinical Trials
Allergan, the maker of Botox, holds close to 800 more patents for potential uses of the drug. It's constantly testing out novel ways of using Botox, and new approvals may come down the line. Some of the conditions the company is working are:
- Erectile dysfunction - researchers believe Botox can provide an erectile boost by thwarting the release of the neurotransmitter norepinephrine, which prevents blood flow. That said, scientific experiments on Botox for erectile dysfunction have been limited.
- Depression- Though many experts are still sceptical, early trials suggest Botox may alleviate symptoms in people with depression. Allergan is currently running trials on Botox efficacy in treating depression.
Final Thoughts
Clinical trials can take years and cost millions of pounds — even billions, So it's no surprise that there are fewer approved treatment areas than there are off-label options. That being said, there's are a lot of off-label uses of Botox that are safe and effective despite the lack of regulatory approval.
Whether you are looking to get either treatment, it's essential you do your own research on the benefits and potential risks. And while you are at, don't forget to verify the credibility and experience of the doctor you want to work with. Remember, even an FDA or MHRA-approved Botox treatment can go wary in the long hands!
Are you interested in a Botox treatment in Birmingham? Talk to a renowned aesthetics doctor about the different approved and off-label Botox treatments and find out which is best for you. Contact us here.